iPS細胞研究
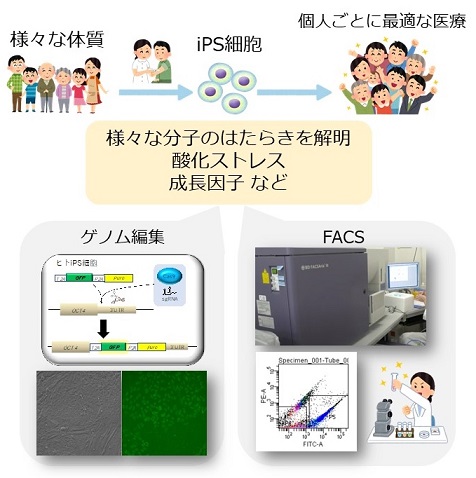
同じように生活していても、人によっては健康だったり、あるいは病気になったりします。このように個人ごとに体質は異なりますが、その差を決めているメカニズムを解明できれば、個人ごとに最適な医療を提供できると考えられます。
ヒトiPS細胞は、そのもとになった個人の体質を反映することが期待されます。そこで私たちはヒトiPS細胞を使って個人差のメカニズムを調べています。ゲノム編集やFACSなどを組み合わせて、最先端の実験を行っています。
 |
|||
| 関連する業績 |
|||
|
Optimized conditions for the supplementation of human-induced pluripotent stem cell cultures with a GSK-3 inhibitor during embryoid body formation with the aim of inducing differentiation into mesodermal and cardiac lineage. Yoda K, Ohnuki Y, Masui S, Kurosawa H, J Biosci Bioeng 129, 371-378 (2020). Optimization of the treatment conditions with glycogen synthase kinase-3 inhibitor towards enhancing the proliferation of human induced pluripotent stem cells while maintaining an undifferentiated state under feeder-free conditions. Yoda K, Ohnuki Y, Kurosawa H, J Biosci Bioeng 127, 381-387 (2019). Srf destabilizes cellular identity by suppressing cell-type-specific gene expression programs. Ikeda T, Hikichi T, Miura H, Shibata H, Mitsunaga K, Yamada Y, Woltjen K, Miyamoto K, Watanabe A, Hiratani I, Yamada Y, Yamamoto T, Hotta A, Okita K, Masui S, Nat Commun 9, 1387 (2018). ROCK inhibitor Y-27632 used during cell aggregate formation adversely affects the early cardiac differentiation on human iPS cells. Ohnuki Y, Kondo T, Kurosawa H, Tissue Cult Res Commun 36, 21-29 (2017). OVOL2 maintains the transcriptional program of human corneal epithelium by suppressing epithelial-to-mesenchymal transition. Kitazawa K, Hikichi T, Nakamura T, Mitsunaga K, Tanaka A, Nakamura M, Yamakawa T, Furukawa S, Takasaka M, Goshima N, Watanabe A, Okita K, Kawasaki S, Ueno M, Kinoshita S, Masui S, Cell Rep 15, 1359-1368 (2016). Effective Rho-associated protein kinase inhibitor treatment to dissociate human iPS cells for suspension culture to form embryoid body-like cell aggregates. Horiguchi A, Yazaki K, Aoyagi M, Ohnuki Y, Kurosawa H, J Biosci Bioeng 118, 588-592 (2014). Transcription factors interfering with dedifferentiation induce cell type-specific transcriptional profiles. Hikichi T, Matoba R, Ikeda T, Watanabe A, Yamamoto T, Yoshitake S, Tamura- Nakano M, Kimura T, Kamon M, Shimura M, Kawakami K, Okuda A, Okochi H, Inoue T, Suzuki A, Masui S, Proc Natl Acad Sci USA 110, 6412-6417 (2013). Effect of hanging drop culture conditions on embryoid body formation and neuronal cell differentiation using mouse embryonic stem cells: Optimization of culture conditions for the formation of well-controlled embryoid bodies. Ohnuki, Y, Kurosawa, H, J Biosci Bioeng, 115, 571-574 (2013). Effect of glucose concentration during embryoid body (EB) formation from mouse embryonic stem cells on EB growth and cell differentiation. Mochizuki H, Ohnuki Y, and Kurosawa H, J Biosc Bioeng, 111, 92-97 (2011). Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells. Masui S, Nakatake Y, Toyooka Y, Shimosato D, Yagi R, Takahashi K, Okochi H, Okuda A, Matoba R, Sharov AA, Ko MSH, Niwa H, Nat Cell Biol 9, 625-635 (2007). Characterization of embryoid bodies of mouse embryonic stem cells formed under various culture conditions and estimation of differentiation status of such bodies. Koike M, Sakaki S, Amano Y, and Kurosawa H, J Biosci Bioeng, 104, 294-299 (2007).査読有 A simple method for forming embryoid body from mouse embryonic stem cells. Kurosawa H, Imamura T, Koike M, Sasaki K, Amano Y, J Biosci Bioeng, 96, 409-411 (2003). |
黒澤研究室HPへのリンク
http://www.ccn.yamanashi.ac.jp/~kurohiro/index.html
